Pancreatic ductal adenocarcinoma (PDAC) remains among the most lethal solid malignancies worldwide, with limited long-term survival despite advances in systemic therapy. A defining biological feature of PDAC is its highly complex tumor microenvironment (TME), which extends beyond malignant epithelial cells to include a dense and heterogeneous stromal compartment.
Read more: Tumor Microenvironment in Pancreatic Cancer Pathogenesis and Therapeutic Resistance
The Desmoplastic Microenvironment in PDAC
PDAC is characterized by an extensive desmoplastic reaction marked by abundant cancer-associated fibroblasts (CAF), altered extracellular matrix (ECM) composition, increased matrix stiffness, and structurally dense stromal architecture. Reduced vascular perfusion and elevated interstitial pressure further contribute to the distinctive biological landscape of this tumor type.
Accumulating evidence indicates that stromal components influence tumor progression, therapeutic response, and tissue architecture. CAF populations exhibit functional heterogeneity and participate in extracellular matrix remodeling, cytokine secretion, and bidirectional signaling with malignant cells. These interactions are widely regarded as central elements in PDAC biology.
Given this complexity, modeling the pancreatic tumor microenvironment in vitro presents substantial experimental challenges.
Limitations of Conventional In Vitro Models
Traditional two-dimensional (2D) pancreatic cancer cell cultures lack three-dimensional structure and fail to recapitulate extracellular matrix constraints or stromal–tumor interactions. Although three-dimensional organoid systems derived from patient tumors more closely reflect epithelial architecture and genetic background, monoculture organoids do not fully incorporate fibroblast-mediated remodeling or stromal signaling dynamics.
These limitations have prompted increasing interest in co-culture systems that integrate CAF populations with patient-derived tumor organoids to better approximate stromal context.
A 3D Pancreatic Cancer Organoid & CAF Co-Culture Platform
In collaboration with clinicians and researchers at Seoul National University Hospital (SNUH), Lambda Biologics established a pancreatic cancer organoid model and developed a co-culture configuration incorporating cancer-associated fibroblasts.
This system was designed to reproduce selected structural and cellular features associated with the pancreatic tumor microenvironment, including epithelial–stromal proximity and matrix-rich architecture. The platform is intended for research use to enable controlled investigation of stromal–tumor interactions within a three-dimensional construct.
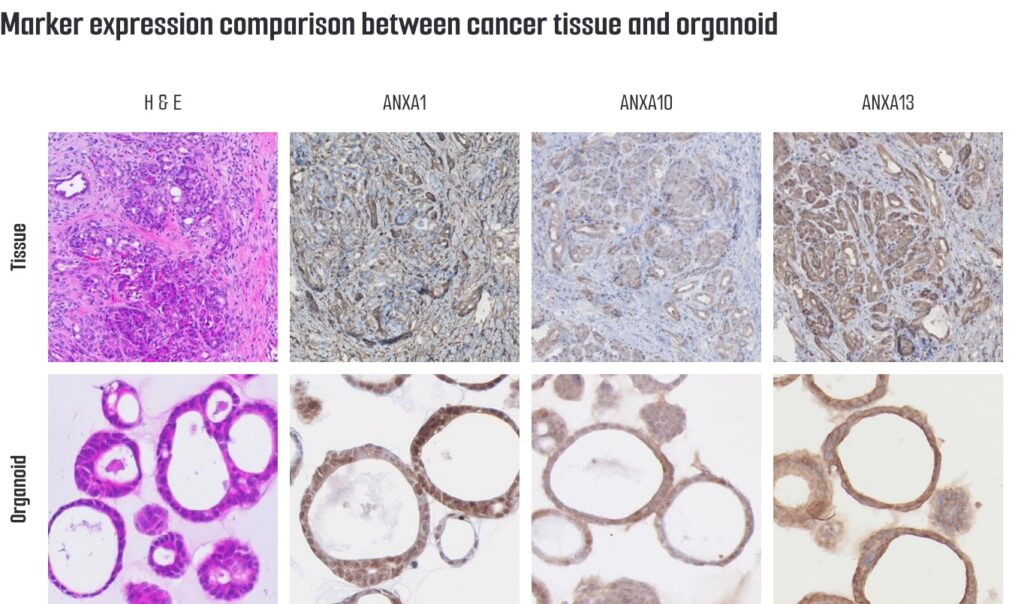
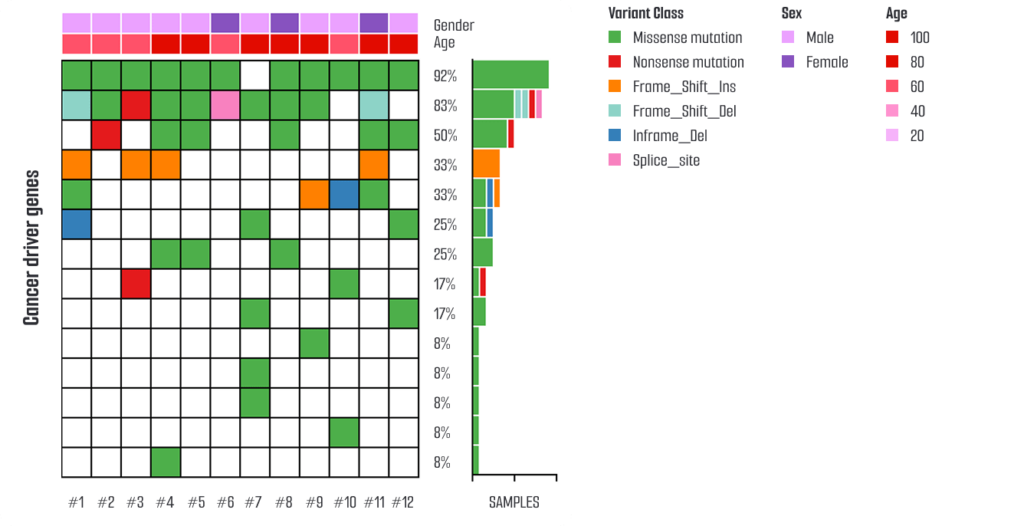
Histological and molecular characterization of the pancreatic organoids demonstrates features consistent with pancreatic tumor phenotypes, including expression of pancreatic cancer–associated markers such as ANXA1, ANXA10, and ANXA13. Whole-exome sequencing (WES) data are available for genetically defined models, supporting mutation-informed mechanistic research.
To better replicate the pancreatic cancer microenvironment, we conducted a collaborative study with Lambda Biologics to establish a tumor organoid model and develop a co-culture platform incorporating cancer-associated fibroblasts (CAF). By leveraging this platform, we aim to accurately assess drug penetration within tumor tissues and facilitate the development of highly effective therapeutic candidates.
Seoul National University Hospital
CAF Integration and Stromal Context
The integration of CAF populations into organoid systems provides a framework for investigating stromal–tumor signaling and extracellular matrix remodeling in vitro. Within such co-culture configurations, researchers may examine how fibroblast-derived factors influence epithelial behavior, structural organization, and molecular pathways.
Three-dimensional stromal incorporation also enables exploratory assessment of compound distribution and diffusion within tumor-like constructs. These applications are intended to generate mechanistic insight under research settings rather than to predict clinical outcomes.
Applications in Translational Oncology Research
Organoid-based systems that incorporate stromal components are increasingly employed in translational oncology research to study tumor microenvironment complexity. In the context of pancreatic cancer, organoid–CAF co-culture platforms may support:
Modeling of stromal-rich tumor architecture in vitro
Investigation of fibroblast-mediated signaling pathways
Analysis of extracellular matrix remodeling dynamics
Mutation-informed exploratory compound response studies
Such systems are designed as experimental tools for preclinical research and are not intended for diagnostic, prognostic, or therapeutic decision-making.
Toward Microenvironment-Aware In Vitro Models
Across oncology research, there is growing emphasis on advanced human-relevant in vitro systems that capture tumor heterogeneity and microenvironmental context. While no in vitro model fully recapitulates the complexity of human tumors, organoid platforms incorporating stromal components represent one approach to addressing limitations inherent to reductionist systems.
In pancreatic cancer research, continued refinement of microenvironment-aware models may contribute to improved mechanistic understanding of tumor–stroma interactions and inform experimental evaluation strategies in preclinical studies.
Lambda Biologics’ Oncology Solutions: Patient-derived cancer organoid-based drug evaluation service
Gastric Cancer Organoid | Breast Cancer Organoid | Hepatocarcinoma Cancer Organoid | Pancreatic Cancer Organoid