Cancer organoid co-culture systems are emerging as powerful tools for evaluating immuno-oncology therapies. By integrating tumor organoids with immune cells such as macrophages and T cells, these models enable researchers to better capture the complexity of the tumor immune microenvironment and generate more physiologically relevant insights for drug development.
Traditional cancer models – including 2D cell lines and many animal systems – often fail to replicate the complex cellular interactions that shape tumor growth and therapeutic response. Organoid-based immune co-culture platforms help bridge this gap by enabling direct investigation of immune-mediated tumor killing and drug response.
Read more: Tumor organoid-immune co-culture models: exploring a new perspective of tumor immunity
In This Article
Modeling the Tumor Immune Microenvironment for Immuno-Oncology Research
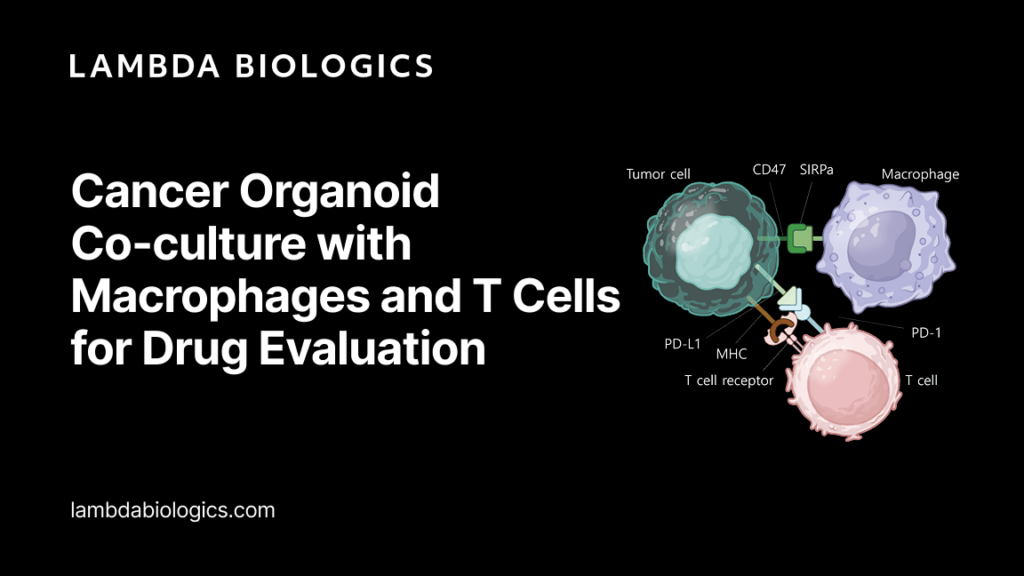
Immune cells are central regulators of tumor progression and therapeutic response. Among them, macrophages and T cells play particularly important roles within the tumor microenvironment.
Macrophages in the Tumor Microenvironment
Macrophages are highly versatile immune cells capable of adapting their functional states in response to environmental signals. Within tumors, macrophages can differentiate into different phenotypes depending on cytokine signaling and local conditions.
Two commonly described macrophage states include:
- M1 macrophages, which exhibit pro-inflammatory properties and are generally associated with anti-tumor activity
- M2 macrophages, which contribute to tissue repair and immune suppression and are often linked to tumor progression
In real tumors, macrophages exist along a spectrum between these states. The balance between M1 and M2 macrophages significantly influences tumor growth, immune regulation, and response to therapy.
T Cells and Cytotoxic Tumor Killing
T cells are another critical component of anti-tumor immunity. Cytotoxic T lymphocytes recognize tumor antigens presented by cancer cells and initiate targeted killing through immune effector mechanisms.
However, the activity of T cells can be strongly influenced by other immune populations, including macrophages. Signals from macrophages can either stimulate or suppress T-cell activation, making it essential to study these interactions within integrated experimental models.
Interested in organoid-based immuno-oncology testing? Contact our scientists to learn more.
A Multi-Immune Cell Organoid Co-culture System by Lambda Biologics
To better capture immune–tumor interactions, Lambda Biologics has developed a cancer organoid co-culture system incorporating macrophages and T cells.
This platform recreates key features of the tumor immune microenvironment by allowing tumor organoids to interact directly with multiple immune cell types within a controlled in vitro setting.
A major advantage of this approach is the ability to observe multiple immune-mediated anti-tumor mechanisms simultaneously, including:
- Macrophage phagocytosis, where macrophages engulf and eliminate tumor cells
- T cell-mediated cytotoxicity, where activated T cells directly kill cancer cells
- Immune signaling interactions between macrophages and T cells that regulate overall anti-tumor responses
By integrating these processes within a single experimental system, the platform provides a more comprehensive understanding of how immune cells cooperate to influence tumor behavior and therapeutic outcomes.
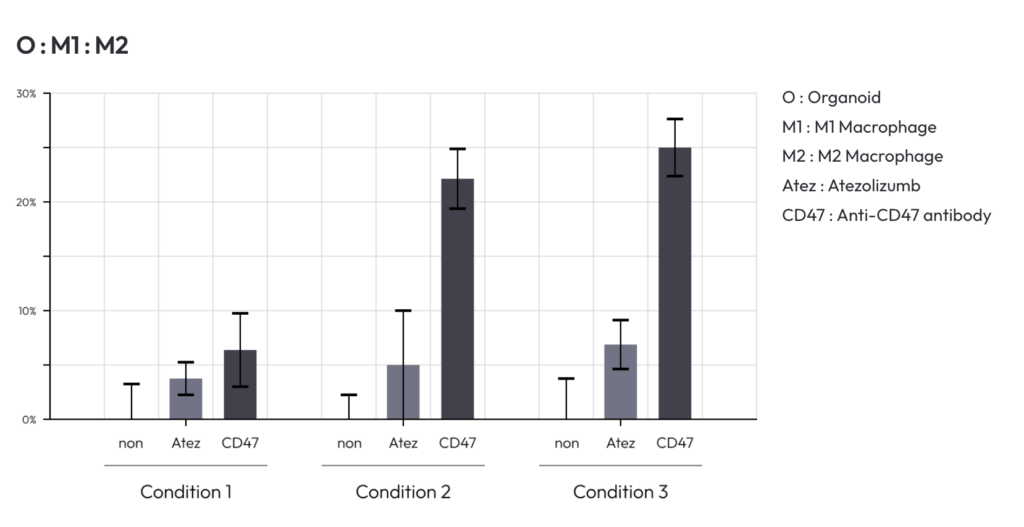
Modeling Macrophage Polarization and Immune Balance
A key feature of the co-culture system is the ability to incorporate both M1 and M2 macrophages.
Because these macrophage phenotypes exhibit distinct immune markers, metabolic characteristics, and gene expression profiles, they perform different roles in tumor biology. The ability to control the ratio of M1 to M2 macrophages allows researchers to model various immune environments that resemble those found in patient tumors.
By adjusting immune cell composition within the culture system, investigators can study how macrophage polarization affects tumor growth and evaluate how candidate therapies influence immune responses.
This capability is particularly important for drugs designed to reprogram tumor-associated macrophages or enhance immune-mediated tumor killing.
Read more: Pancreatic Cancer Organoid Co-Culture with CAF for Tumor Microenvironment Modeling
Optimizing Drug Evaluation with Organoid Co-culture Platforms
Developing a reliable multi-cellular model requires careful optimization of experimental conditions.
Within the organoid co-culture platform, different ratios of tumor organoids, macrophages, and T cells can be tested to identify conditions that best replicate tumor immune dynamics. Drug and antibody treatments are then evaluated under these optimized settings.
Key experimental readouts include:
- Tumor organoid growth rates
- Tumor cell death and cytotoxicity
- Immune cell activity and interactions
By analyzing these parameters, researchers can establish robust evaluation criteria for assessing the effectiveness of immuno-oncology therapies.
Stem Cell-Derived Macrophages for Scalable Testing
Another important feature of the platform is the use of macrophages derived from pluripotent stem cells.
Primary macrophages obtained from human donors can vary significantly between samples and are often limited in supply. Stem cell-derived macrophages provide a more standardized and scalable alternative for experimental testing.
These cells support:
- Improved experimental reproducibility
- Consistent immune cell populations
- Efficient large-scale drug screening
As a result, stem cell-derived macrophages enable the development of reliable and scalable immune-organoid co-culture systems suitable for drug discovery applications.
New Updates: Lambda Biologics’ Macrophage-Driven Drug Evaluation Platforms
To support diverse research applications, Lambda Biologics has developed specialized co-culture platforms for evaluating macrophage-driven immune responses in cancer.
Monocyte-derived Macrophage Co-culture Platform
The model focuses on macrophage-targeted anticancer drug evaluation using monocytes isolated from autologous or HLA-matched blood samples. These monocytes are differentiated into macrophages and cultured with tumor organoids within an extracellular matrix (ECM)-based system. This configuration enables direct comparison of cytokine-driven M1 and M2 macrophage differentiation and the resulting cytotoxic effects on tumor organoids.
Activated T Cell Co-culture Platform Induced by Monocyte-derived Macrophages
A second model extends this system by incorporating T-cell activation mediated by macrophages. In this platform, macrophages derived from autologous or HLA-matched monocytes stimulate T-cell activation, which subsequently contributes to tumor organoid cytotoxicity. This setup allows researchers to evaluate both macrophage-driven and T-cell-mediated immune responses, providing deeper insight into the mechanisms of immuno-oncology therapies.
Both platforms are built on an ECM-based organoid culture system designed to replicate the structural and signaling environment of human tumors, enabling more physiologically relevant drug evaluation.
Read more: Tumor Organoids in Drug Discovery: Applications, Advantages, and Limitations
Advancing Immuno-Oncology Drug Discovery
As immuno-oncology continues to evolve, predictive preclinical models are becoming increasingly important. Organoid-based immune co-culture systems offer a powerful approach for studying complex tumor–immune interactions and evaluating therapeutic responses in a controlled laboratory setting.
By integrating tumor organoids with macrophages and T cells, Lambda Biologics oncology platforms provide a more physiologically relevant framework for assessing immuno-oncology drugs and antibodies. Ultimately, such models have the potential to accelerate drug discovery and improve the translation of preclinical findings into effective cancer therapies.
Lambda Biologics’ Oncology Solutions: Patient-derived cancer organoid-based drug evaluation service
Gastric Cancer Organoid | Breast Cancer Organoid | Hepatocarcinoma Cancer Organoid | Pancreatic Cancer Organoid





