A recent article published in Nature (2026) highlights a significant global shift: the gradual transition away from animal experimentation toward human-relevant scientific models. The UK has announced a phased plan to eliminate certain animal tests, while in the United States, the U.S. Food and Drug Administration (FDA) and the National Institutes of Health (NIH) are moving to make animal studies the exception rather than the norm in drug safety assessment. The European Commission is also preparing a roadmap to end animal testing in chemical safety evaluations.
This momentum is driven by both ethical considerations and rapid advances in New Approach Methodologies (NAMs), including organs-on-chips, 3D organoids, and computational or AI-based models. Built using human cells and clinical data, these systems aim to improve predictive accuracy – particularly important given that approximately 86% of investigational drugs fail during clinical trials.
However, as Nature emphasizes, NAMs are not yet positioned to fully replace animal models. Many biological systems remain too complex to replicate in vitro, and rigorous validation is essential before regulators broadly accept alternative data. The transition, therefore, is expected to be staged, evidence-based, and closely aligned with regulatory standards.
Research article: The age of animal experiments is waning. Where will science go next?
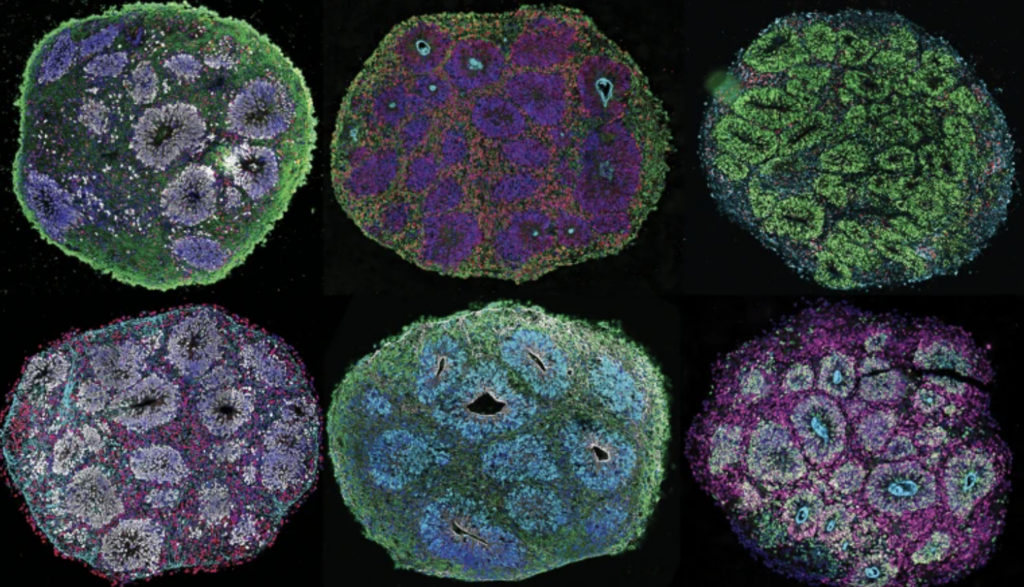
Lambda Biologics’ Organoid Solutions: Advancing Ethical Science, Together
Drug development demands accuracy, speed, and relevance to human biology. At Lambda Biologics, we deliver advanced organoid-based platforms that replicate the complexity of human organs – empowering pharmaceutical and biotechnology companies to make better decisions, faster.
Cerebral Organoid | Midbrain Organoid | Skin & Hair Organoid | Intestine Organoid |