Cancer treatment has long faced a fundamental dilemma: the drugs powerful enough to kill tumors are often too toxic to spare healthy tissue. Chemotherapy can’t tell a cancer cell from a heart cell – which is why patients endure hair loss, immune compromise, and organ damage alongside treatment. Monoclonal antibodies brought precision, but precision alone doesn’t kill – many aren’t potent enough to eradicate aggressive tumors on their own.
Antibody-drug conjugates (ADCs) solve both problems at once: a highly potent drug attached directly to an antibody, delivered right where it needs to be. This article explains what ADCs are, how they work, and what it takes to develop them successfully.

In This Article
ADCs – A Simple Definition
Antibody-drug conjugates (ADCs) are a class of targeted cancer therapies that combine a monoclonal antibody with a cytotoxic payload via a chemical linker. The antibody guides the drug precisely to cancer cells that overexpress a specific antigen, while the payload delivers a lethal cytotoxic dose upon internalization – sparing surrounding healthy tissue in a way conventional chemotherapy cannot.
As of 2025, the FDA has approved 15 ADCs, with two new approvals granted in 2025 alone: datopotamab deruxtecan (Dato-DXd) and telisotuzumab vedotin (Emrelis). More than 100 additional candidates are currently in active clinical development.
The Core Insight Behind ADCs
ADCs were designed to combine the navigational precision of a monoclonal antibody with the destructive power of a cytotoxic payload – delivering a lethal dose directly inside tumor cells while sparing surrounding healthy tissue. Think of them as a guided missile: the antibody is the guidance system; the payload is the warhead.
The Three Key Components of an ADC
Think of an ADC as a guided missile: the antibody is the guidance system, the linker is the safety pin, and the payload is the warhead.
1. The Antibody – The Targeting System
The antibody’s job is to find and latch onto cancer cells – specifically, cells that display an abnormally high amount of a particular protein (called an antigen) on their surface. Healthy cells typically express little to none of that same protein, which is what makes this targeting possible.
Once the antibody locks onto its target, the whole ADC complex is pulled inside the cancer cell – triggering the next step. Common targets in approved ADCs include HER2 (breast, gastric, and lung cancers), TROP2, CD30, CD33, and Nectin-4.
2. The Linker – The Safety Pin
The linker keeps the toxic payload firmly attached to the antibody during circulation – preventing it from leaking out into healthy tissue. Once inside a cancer cell, the linker is broken down by the cell’s own internal environment, releasing the payload exactly where it needs to be.
There are two types: cleavable linkers (broken down by enzymes or acidity inside the cell) and non-cleavable linkers (which require the entire antibody to be degraded first). Cleavable linkers have an added advantage – the released payload can sometimes “spill over” into neighboring cancer cells, killing even those that the antibody couldn’t directly reach.
3. The Payload – The Warhead
The payload is an ultra-potent cell-killing drug – up to 1,000 times more toxic than conventional chemotherapy. That extreme potency is intentional: only a small amount of payload reaches the tumor, so it needs to be powerful enough to be lethal in tiny doses.
Payloads work by either damaging DNA (preventing the cell from replicating) or disrupting cell division (locking the cell in a state where it can no longer divide). A newer class – topoisomerase I inhibitors like DXd, the payload in Enhertu – is behind several of the most recently approved ADCs.
A Note on DAR (Drug-to-Antibody Ratio)
DAR measures how many payload molecules are attached to each antibody. Too few and the ADC isn’t potent enough; too many and it becomes unstable and clears too quickly from the body. Most approved ADCs are engineered to carry 2–4 payload molecules per antibody – a balance refined through decades of development.
How Does an ADC Work
After being administered by IV infusion, an ADC travels through the bloodstream fully intact – the payload stays locked to the antibody until it reaches the right destination. Here’s what happens next:
- Seek: The antibody scans for cancer cells displaying its specific target protein. When it finds one, it locks on.
- Enter: The cancer cell “swallows” the entire ADC, pulling it inside through its normal membrane recycling process.
- Disarm: Inside the cell, an acidic compartment called the lysosome acts like a chemical shredder – breaking down the linker and releasing the payload.
- Destroy: The free payload floods the cell interior, either shredding its DNA or jamming its ability to divide. The cell dies.
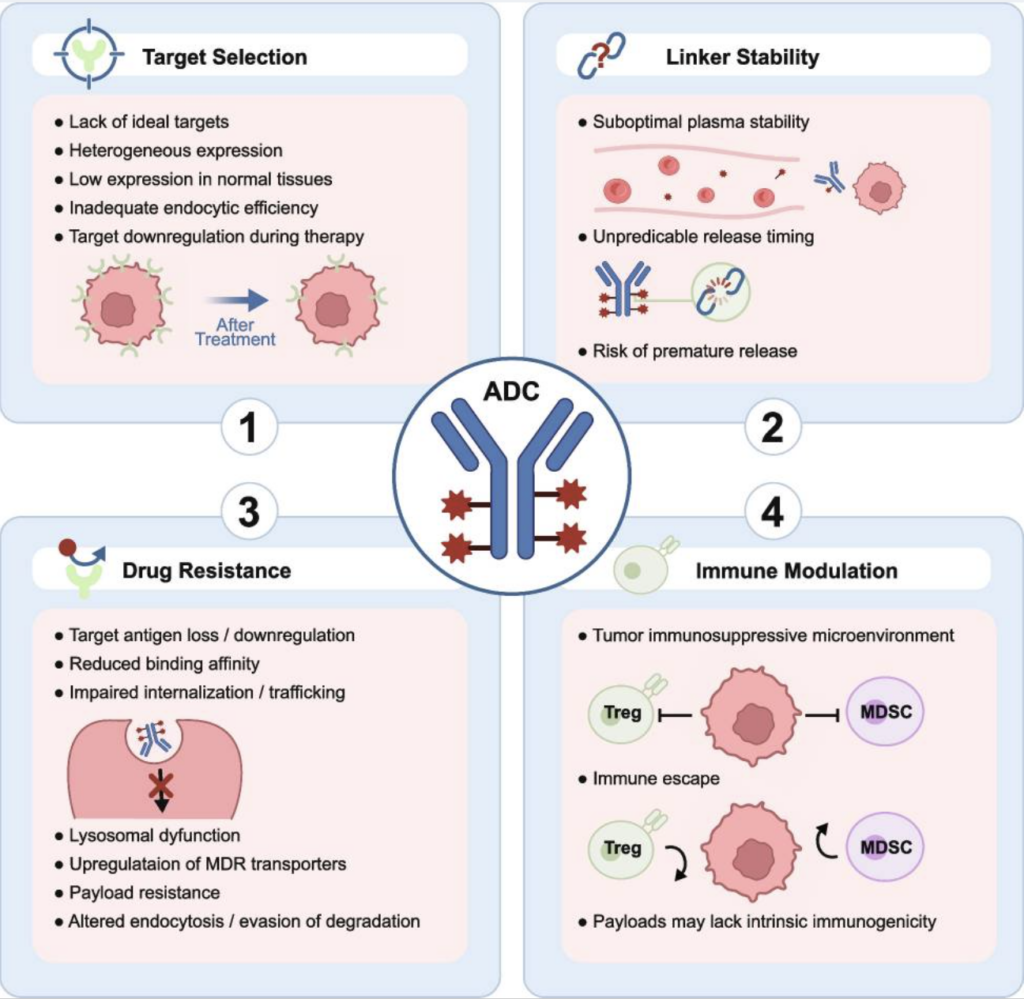
Key Challenges in ADC Development
Despite their clinical promise, ADCs remain technically demanding to develop. Researchers must contend with:
- Antigen heterogeneity: Uneven expression of target antigens within a tumor can leave subpopulations of cancer cells unaffected.
- Drug penetration in solid tumors: Large ADC molecules struggle to reach deep into solid tumors, which are often surrounded by dense, fibrous tissue that acts as a physical barrier to drug delivery.
- Narrow therapeutic window: The extreme potency of ADC payloads demands tight control of DAR, linker stability, and antigen selectivity to avoid off-target toxicity.
- Resistance mechanisms: Cancer cells can evade ADCs through antigen downregulation, impaired internalization, altered lysosomal trafficking, or drug efflux pumps that actively expel the payload before it can act.
Why Preclinical Model Choice Matters
Many ADC failures in clinical trials can be traced back to limitations in preclinical testing. Traditional 2D cell cultures lack tumor architecture and stromal interactions; mouse xenograft models introduce species-specific differences in antigen expression and immune biology. More physiologically relevant models – such as patient-derived tumor organoids – are increasingly recognized as essential tools for predicting ADC efficacy, penetration, and resistance in a human tumor context.
Lambda Biologics: Organoid Platforms for ADC Research
Lambda Biologics is a Leipzig-based company specialized in patient-derived tumor organoid (PDO) technology for oncology drug development. Through its ODISEI ONC platform, Lambda provides 3D organoid models derived directly from patient tumor tissue, designed to preserve native genetic heterogeneity, antigen expression patterns, and tumor microenvironment (TME) components – precisely the variables that determine ADC behavior in vivo.
Lambda Biologics’ capabilities relevant to ADC research include:
- Patient-derived cancer organoids from colorectal, breast, gastric, pancreatic, hepatocellular, lung and more – aligned with major ADC target indications
- TME co-culture systems integrating cancer-associated fibroblasts (CAFs), macrophages, and T cells to model stromal barriers and immune interactions affecting ADC penetration and efficacy
- Spatial biology and molecular profiling (PhenoCycler, CosMx, scRNA-seq) for high-resolution mapping of antigen expression and drug distribution within organoid tissue
- Animal-free, human-relevant testing aligned with New Approach Methodologies (NAMs), reducing species-specific translational gaps inherent in murine preclinical models
If you are developing ADC candidates and need preclinical models that more accurately reflect the biology of your target tumor, contact Lambda Biologics to explore how our ODISEI ONC platform can support your pipeline.
Conclusion
Antibody-drug conjugates represent a genuine paradigm shift in oncology – a therapeutic class that combines molecular precision with cytotoxic potency in a way that neither antibodies nor conventional chemotherapy can achieve alone. With 15 FDA approvals and over 100 candidates in clinical development, ADCs are now a central pillar of the precision oncology toolkit.
Yet the complexity that makes ADCs so promising also makes them technically demanding to develop. Antigen heterogeneity, solid tumor penetration barriers, narrow therapeutic windows, and resistance mechanisms mean that the quality of preclinical decision-making – including the models used to generate it – directly influences clinical outcomes.
The question is no longer whether ADCs work – the clinical data is clear. The question is how to develop them faster, smarter, and with fewer late-stage failures. That starts with better models.
Research articles:
Antibody–drug conjugates in cancer therapy: current landscape, challenges, and future directions
Lambda Biologics’ Oncology Solutions: Patient-derived cancer organoid-based drug evaluation service
Gastric Cancer Organoid | Breast Cancer Organoid | Hepatocarcinoma Cancer Organoid | Pancreatic Cancer Organoid


