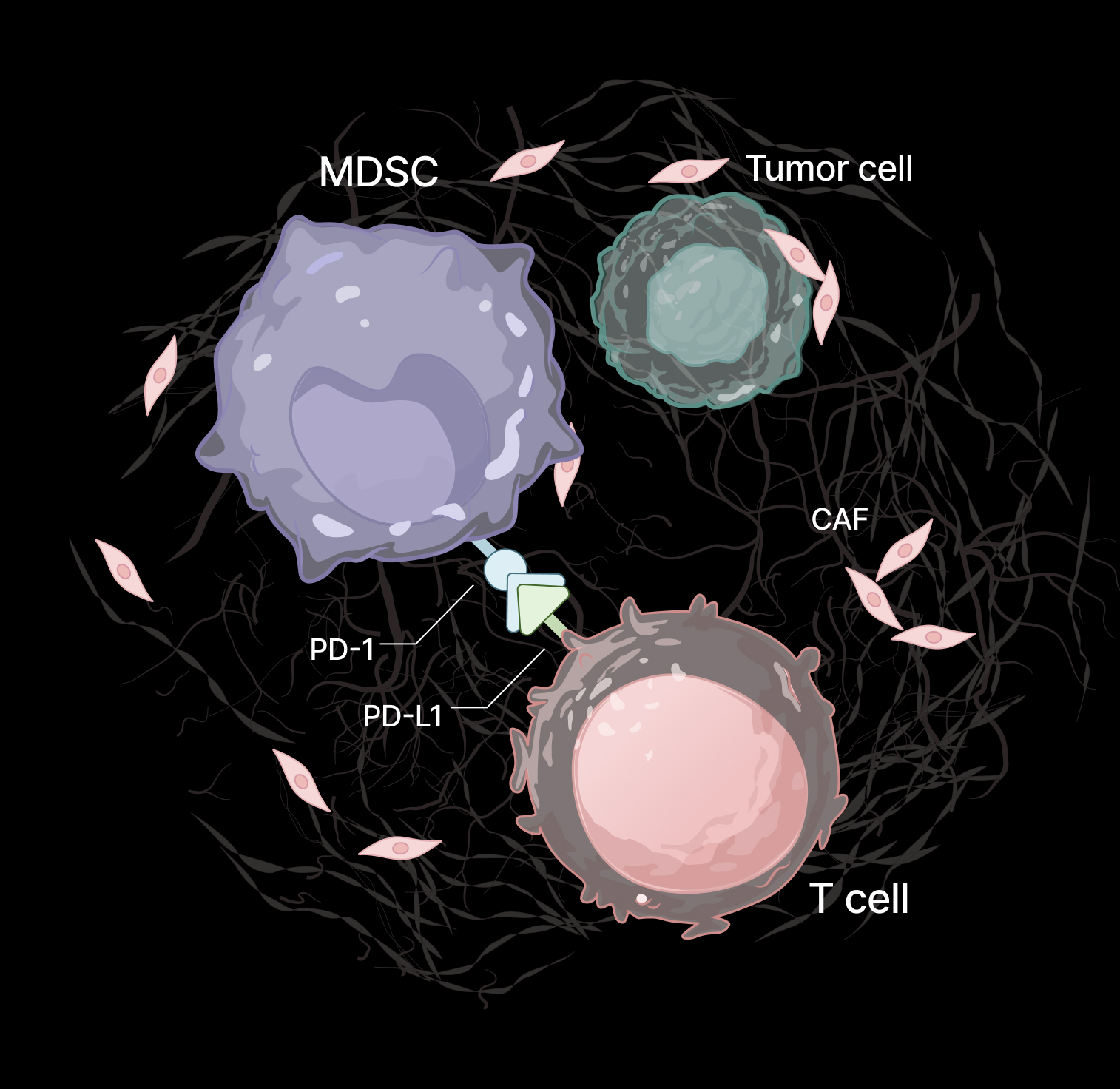
MDSCs migrate toward tumor-derived signals and infiltrate tumor tissues through extracellular matrix barriers. The Transwell system models this process by enabling measurement of directional migration and invasion toward tumor organoids and CAFs.
MDSCs suppress T-cell activation and clustering through both direct cell–cell interactions and soluble factors. Functional co-culture assays enable quantification of this immunosuppressive activity.
The inclusion of both tumor organoids and CAFs enables modeling of synergistic signaling within the TME, which is critical for accurately reproducing MDSC recruitment dynamics.
THP-1 cells are first differentiated under cytokine-driven conditions to generate MDSC-like cells. These cells are validated to exhibit an MDSC-consistent phenotype through flow cytometry and to express immune-regulatory gene signatures as confirmed by qRT–PCR, while remaining distinct from conventional macrophage differentiation states.
Their immunosuppressive function is then assessed using PBMC and T-cell co-culture assays, where MDSC-like cells demonstrate the ability to suppress T-cell clustering through both contact-dependent interactions and soluble factor–mediated mechanisms.
For the co-culture system, MDSC-like cells are seeded in the upper chamber of a Matrigel-coated Transwell insert with a 5 µm pore size, while colorectal cancer (CRC) organoids , CAFs, or their combination are placed in the lower chamber. This configuration enables the assessment of MDSC trafficking and infiltration under defined multicellular conditions.
Candidate compounds can be introduced into the system to evaluate their effects on MDSC behavior, including both direct cellular responses and changes in migration and infiltration within the co-culture setup.
MDSC immunosuppressive function is assessed through T-cell clustering assays, while trafficking and infiltration are quantitatively measured using imaging-based cell counting and flow cytometry (FACS), providing orthogonal and complementary readouts. The platform demonstrates that tumor organoids and CAFs contribute to MDSC infiltration dynamics, while MDSC-like cells exhibit robust immunosuppressive activity in co-culture systems.
In drug profiling studies, differential responses are observed across tumor organoids, MDSC-like cells, and CAFs, enabling evaluation of cell type–specific effects. Importantly, certain compounds reduce MDSC-mediated immunosuppression, while others modulate MDSC trafficking and infiltration in a concentration-dependent manner, highlighting distinct and complementary therapeutic strategies.
Ultimately, it supports the development of next-generation immunotherapies aimed at reprogramming the tumor microenvironment and overcoming resistance.