
iPSC Generation Services deliver quality-controlled, integration-free induced pluripotent stem cells for human-relevant research. With over 1,000 iPSC lines generated, Lambda Biologics applies standardized workflows and rigorous quality control, including RNA reprogramming vector clearance and mycoplasma testing. Using Sendai virus–based, non-integrating reprogramming, we produce transgene-free iPSCs suitable for downstream applications such as disease modeling, drug discovery, toxicity testing, and organoid development.
Price | 5146€+ |
Organism | Human |
Product Type | iPSC |
Tissue | Kreatinocytes, Urine, Blood, PBMCs |
Disease | – |
Applications
Toxicity
Organoid Based

Advancing K-Beauty with Skin Organoids: A Next-Generation Platform for Non-Animal Testing and High-Precision Cosmetic Innovation With the global rise of K-beauty, the cosmetics industry continues to grow steadily. Since the...

Traditional microscopy methods often require fluorescent labeling to analyze cellular structures, which can be time-consuming and invasive. In contrast, our HT-X1 system allows for high-resolution visualization of cellular morphology without...

Traditional protein analysis has primarily focused on quantifying expression levels within tissue samples. However, recent advances in spatial analysis techniques have shifted attention toward evaluating not only expression levels, but...
Among the many fermented foods we consume, kimchi is particularly known for containing a diverse range of lactic acid bacteria, which are believed to influence the activation of immune cells...
We conducted a study focused on identifying disease-related markers using patient-derived tissue samples. However, traditional methods limited our ability to analyze multiple candidate markers simultaneously, and the limited availability of...
Induced pluripotent stem cells (iPSCs) are created by reprogramming somatic cells to gain pluripotency, allowing them to differentiate into any cell type in the body.
This capability is crucial for studying cellular functions, creating disease models, and developing patient-specific therapies.
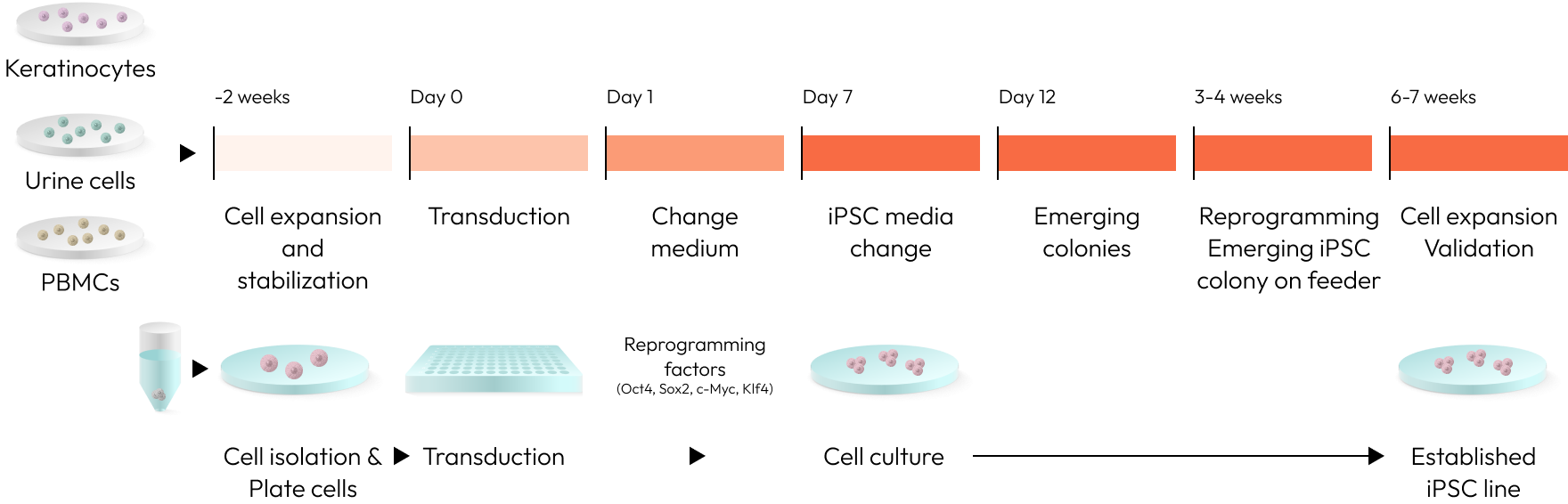
We utilize a variety of cells, including PBMC (peripheral blood mononuclear cells) and fibroblasts, to produce iPSCs.
Notably, our iPSC production method employs a non-integrating approach, ensuring both safety and efficiency.
By generating iPSCs without genetic modification, we obtain more natural and stable stem cells.
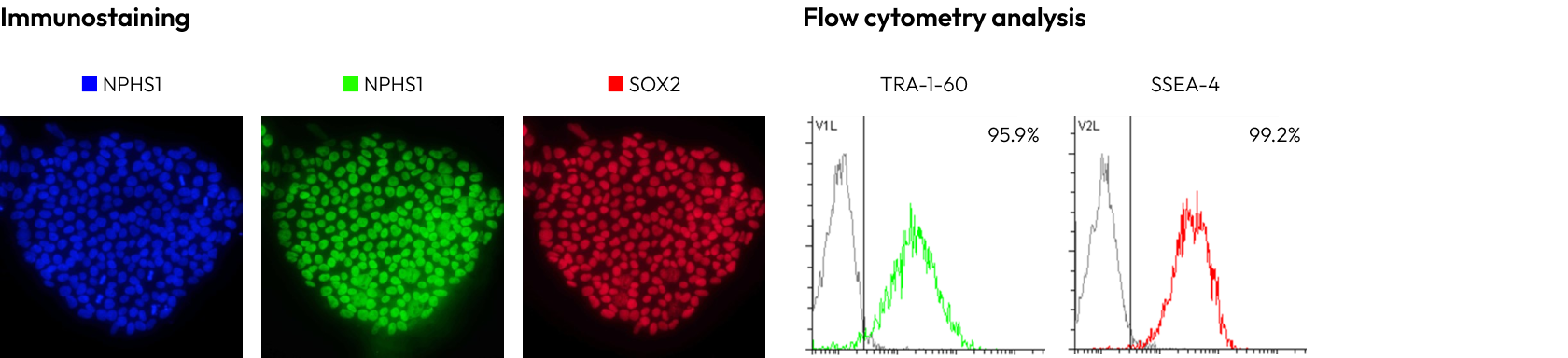
We rigorously check the presence of key pluripotency markers via flow cytometry and immunocytochemistry (ICC) to ensure that iPSCs remain undifferentiated until needed for your specific research purposes.
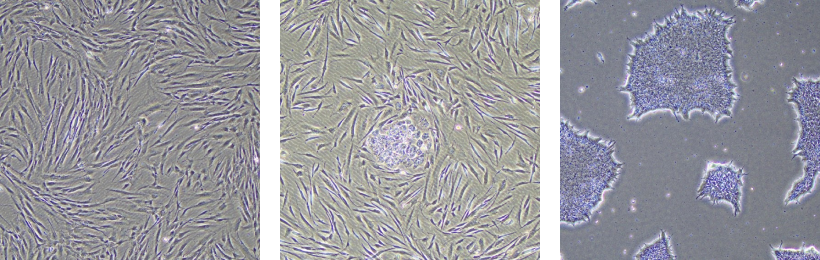
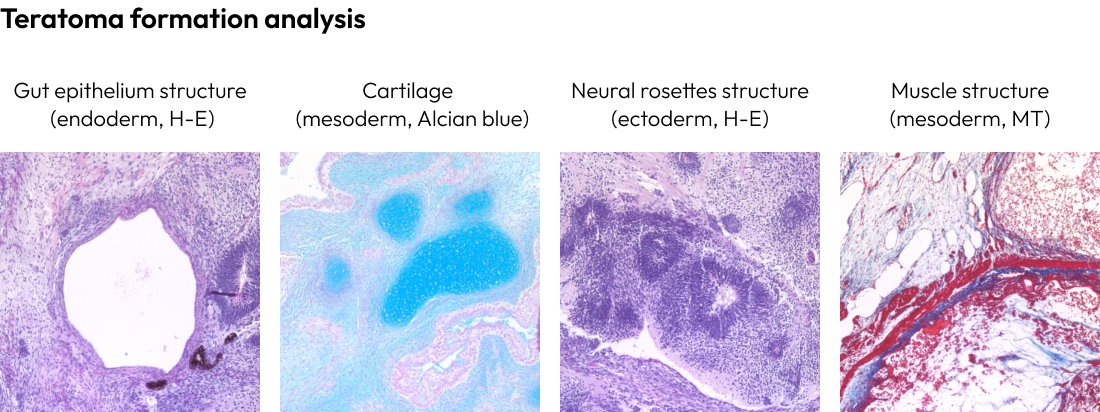
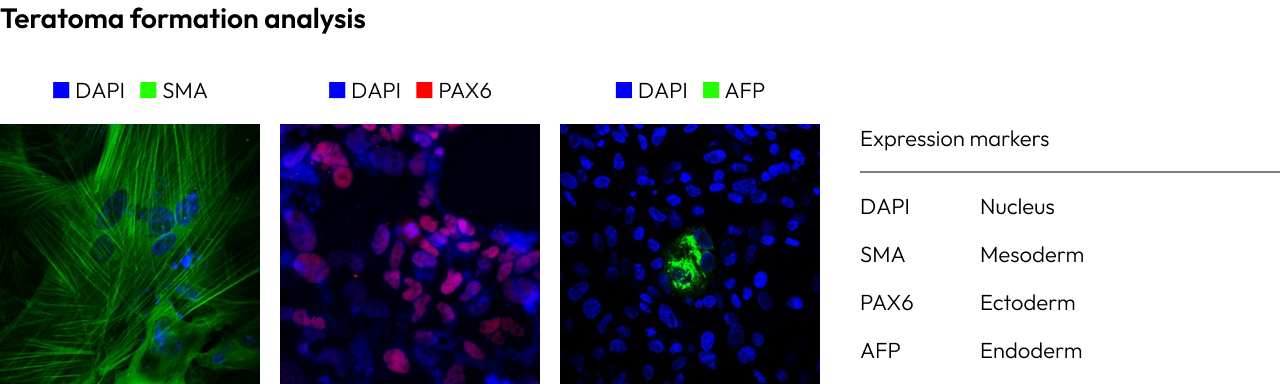
Verifying the true pluripotency of iPSCs (induced pluripotent stem cells) is a critical step in research.
The confirmation of trilineage differentiation involves assessing the ability of iPSCs to differentiate into the three germ layers: endoderm, mesoderm, and ectoderm.
This evaluation is conducted through teratoma formation and spontaneous differentiation into the three germ layers.
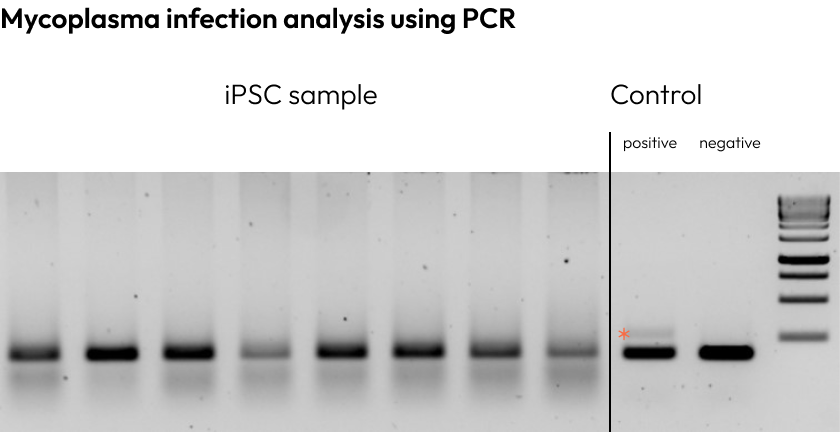
To guarantee safety of our cell cultures, we perform thorough mycoplasma contamination testing.
This ensures that our iPSCs are free from contaminants that could compromise your research.
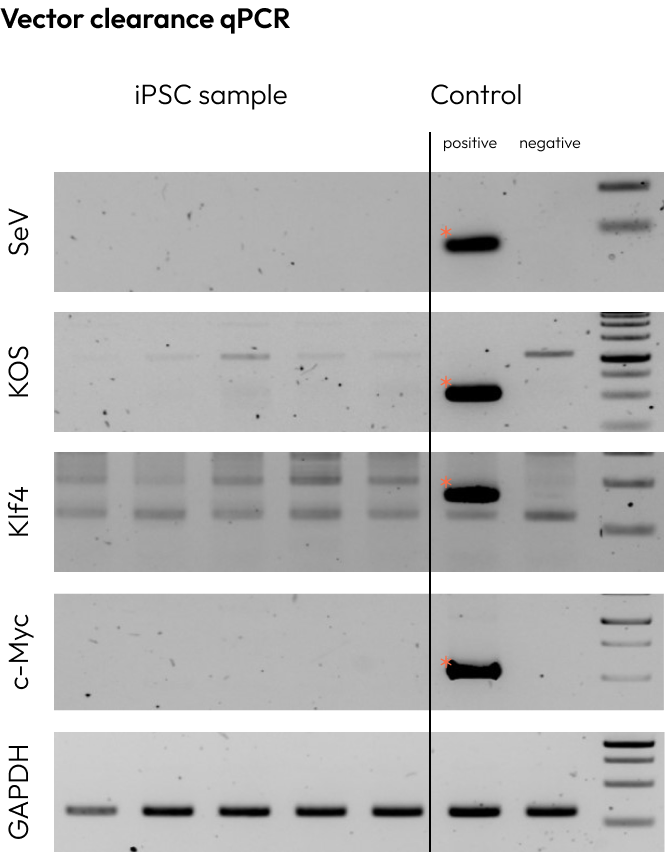
We confirm the removal of reprogramming vectors to prevent any residual vector sequences from affecting the iPSCs
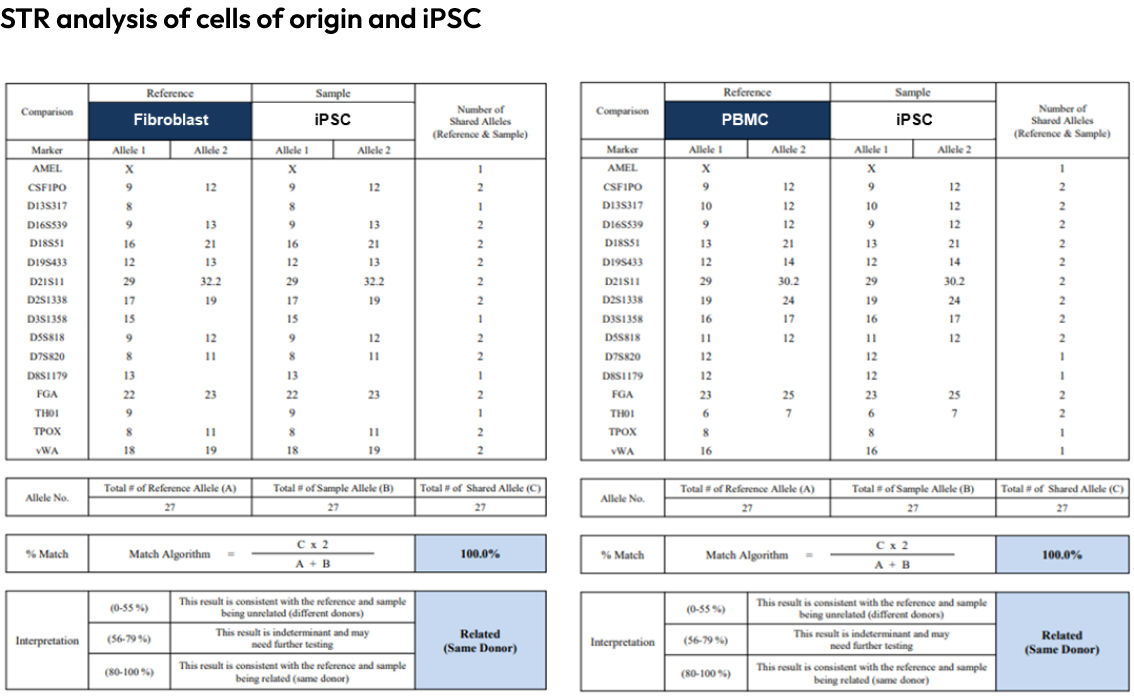
STR analysis is used to confirm that the original somatic cells and the reprogrammed iPSCs share the same genetic profile.
This ensures that the cells have been correctly reprogrammed.
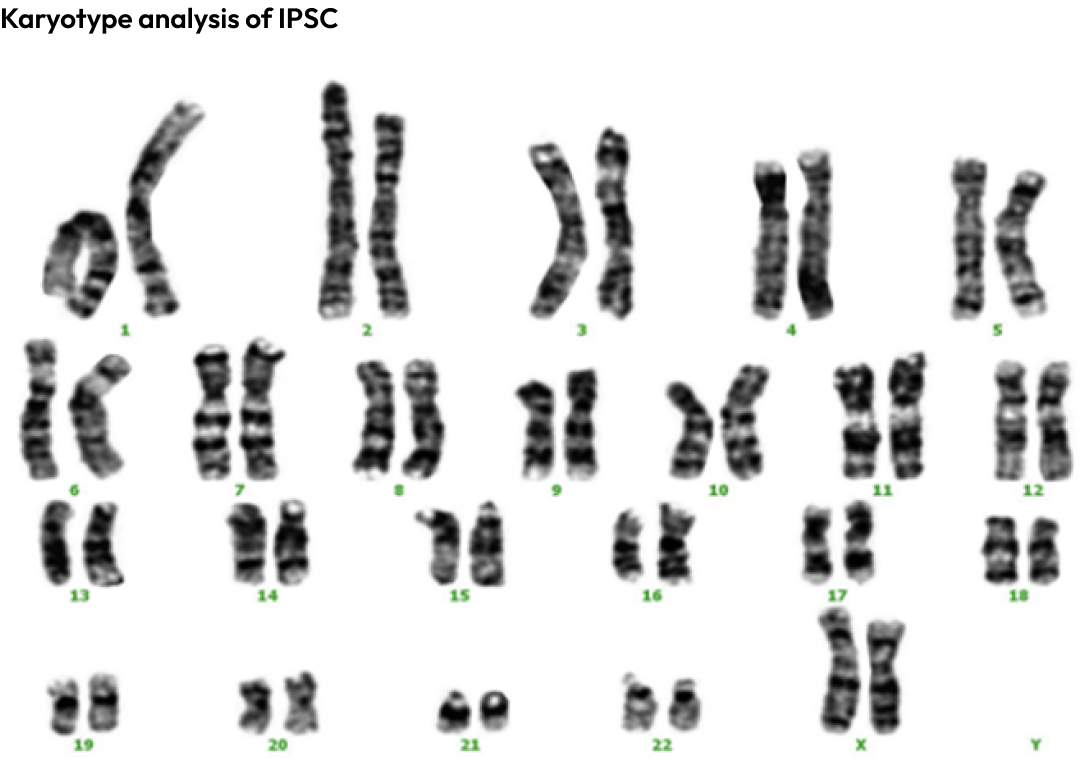
Through karyotype analysis, it was confirmed that there were no chromosomal mutations in the iPSCs produced.
Planning to use quality-controlled iPSCs in your research?
Get in touch with Lambda Biologics to discuss your scientific goals and requirements.
An iPSC generation service reprograms human somatic cells into induced pluripotent stem cells (iPSCs) that can self-renew and differentiate into multiple cell types. These iPSCs provide a human-relevant platform for disease modeling, drug discovery, toxicity testing, and advanced 3D systems such as organoids.
We accept commonly used human somatic cell sources, including peripheral blood mononuclear cells (PBMCs) and skin fibroblasts. Samples should meet basic viability and cell number requirements. Detailed submission guidelines and pre-assessment support are provided to ensure efficient and reproducible reprogramming.
All generated iPSC lines undergo rigorous quality control, which may include:
This ensures suitability for downstream research and translational applications.
iPSCs can be used across a wide range of applications, including:
Lambda Biologics combines integration-free iPSC generation with deep expertise in human-relevant in vitro and organoid models. We offer reliable quality control, flexible project customization, and seamless integration with downstream differentiation and assay services – supporting research from early discovery to advanced translational studies.