Lambda Biologics congratulates our partner ORGANOIDSCIENCES on a landmark achievement: IND approval from Korea’s Ministry of Food and Drug Safety (MFDS) for ATORM-C – making it the world’s first organoid-based regenerative therapy to enter clinical trials.
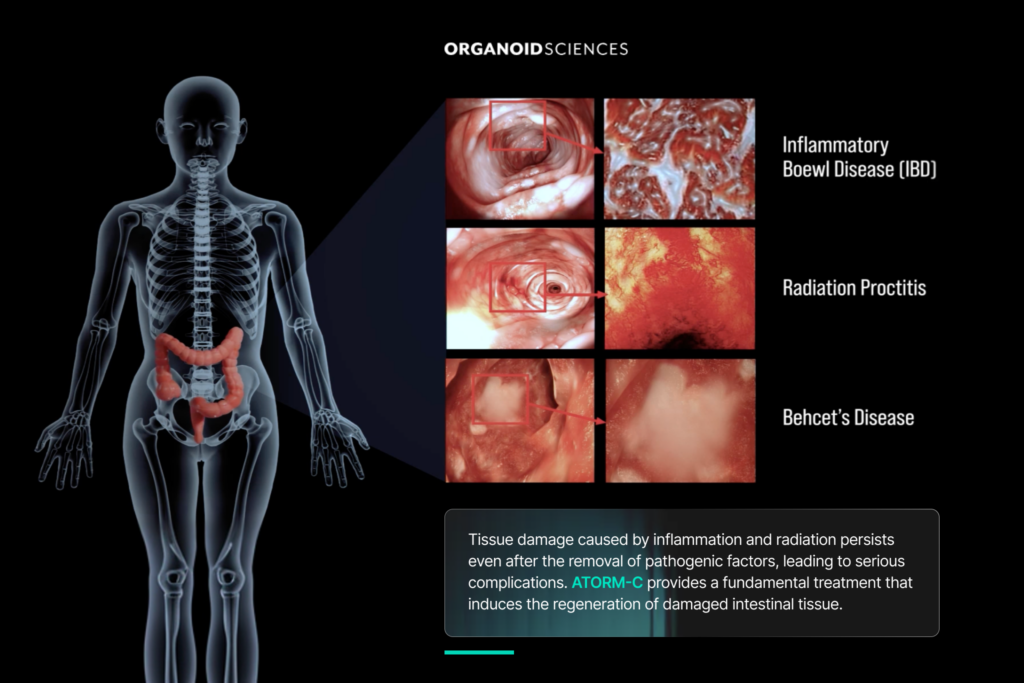
ATORM-C is an intestinal organoid therapy composed of three-dimensional organoids derived from tissue-extracted stem cells from the patient’s own intestinal tissue. By promoting regeneration of damaged intestinal mucosal tissue, it targets refractory ulcers unresponsive to existing therapies – reducing intestinal inflammation and preventing disease progression. Upon transplantation into the damaged mucosa, ATORM-C differentiates into actual intestinal epithelial cells, directly inducing tissue regeneration.

Crohn’s disease is a chronic inflammatory bowel disorder characterized by recurring inflammation and ulcers in the intestinal mucosa. If left untreated, it can progress to severe complications including intestinal strictures and perforation – yet a significant number of patients continue to experience persistent ulcers despite current treatments.
The Phase 1 trial at Asan Medical Center in Seoul will assess tolerability, safety, maximum tolerated dose (MTD), and recommended Phase 2 dose (RP2D), alongside exploratory efficacy. A total of 9–18 patients will be monitored for up to ~24 weeks following administration.
This approval reflects the maturation of organoid technology from research tool to clinical reality – a transition Lambda Biologics is proud to support. In 2024, our collaboration with ORGANOIDSCIENCES was awarded public funding from the Free State of Saxony, backed by the European Union, to accelerate organoid-based therapies within the ATMP framework.
As organoids move from bench to bedside, we remain committed to scaling the human-relevant technologies that will define the next generation of regenerative medicine. Learn more about our organoid partnerships or contact us to explore collaboration opportunities.
Learn more:
Lambda Biologics’ Organoid Solutions: Advancing Ethical Science, Together
Drug development demands accuracy, speed, and relevance to human biology. At Lambda Biologics, we deliver advanced organoid-based platforms that replicate the complexity of human organs – empowering pharmaceutical and biotechnology companies to make better decisions, faster.
Cerebral Organoid | Midbrain Organoid | Skin & Hair Organoid | Intestine Organoid |
FAQs
What is ATORM-C?
ATORM-C is an autologous regenerative therapy developed by ORGANOIDSCIENCES, composed of intestinal organoids derived from the patient’s own tissue-extracted stem cells. Using three-dimensional organoid culture technology, it is directly transplanted into damaged intestinal mucosa, where it differentiates into intestinal epithelial cells to regenerate damaged tissue and treat refractory ulcers unresponsive to existing therapies.
Why is ATORM-C significant for Crohn's disease treatment?
Crohn’s disease is a chronic inflammatory bowel condition where a significant number of patients continue to experience persistent ulcers despite current treatments. ATORM-C represents a fundamentally new approach – rather than managing symptoms, it targets tissue regeneration directly at the source, offering new therapeutic possibilities for patients who do not adequately respond to conventional therapies.
What is the role of organoids in regenerative medicine?
Organoids are miniaturized, three-dimensional tissue models grown from human stem cells that closely replicate the structure and function of real organs. In regenerative medicine, they serve as both a powerful research tool and, increasingly, a therapeutic platform – as demonstrated by ATORM-C’s landmark entry into clinical trials, the first of its kind in the world.





